FRI Biomedical Chemistry Research Stream
From Biomolecules to Treatments—Improving Health at the Human Level by Investigating Cells at the Molecular Level
The research carried out by first-year students in the Biomedical Chemistry research stream at Binghamton University contributes to better understanding of how to improve targeted delivery of biomedicine in the human body. In particular, the stream research focuses on understanding the role and requirement of biomolecules in neurological disorders and cancer. Researchers within this steam will identify molecular targets for therapeutic treatment.
Biomedical Chemistry research intersects the traditional disciplines of Pharmacology, Chemistry, Genetics, Biology, Medicinal Chemistry, Cellular Biology, Neurochemistry, and Molecular Biology. The research questions our FRI students explore are to investigate biomolecules in neurodegenerative diseases and cancer to better understand how they function and, in turn, provide insight in targeting and designing novel medicinal therapies.
All of the research projects in this field will advance medicinal practices. Each discipline, neurochemistry, genetics, molecular and cellular biology, biophysics, medicinal chemistry, and pharmacology, is intertwined to allow for an increased understanding of cellular complexities and will aid in developing therapeutic avenues to support public health.
Research Themes
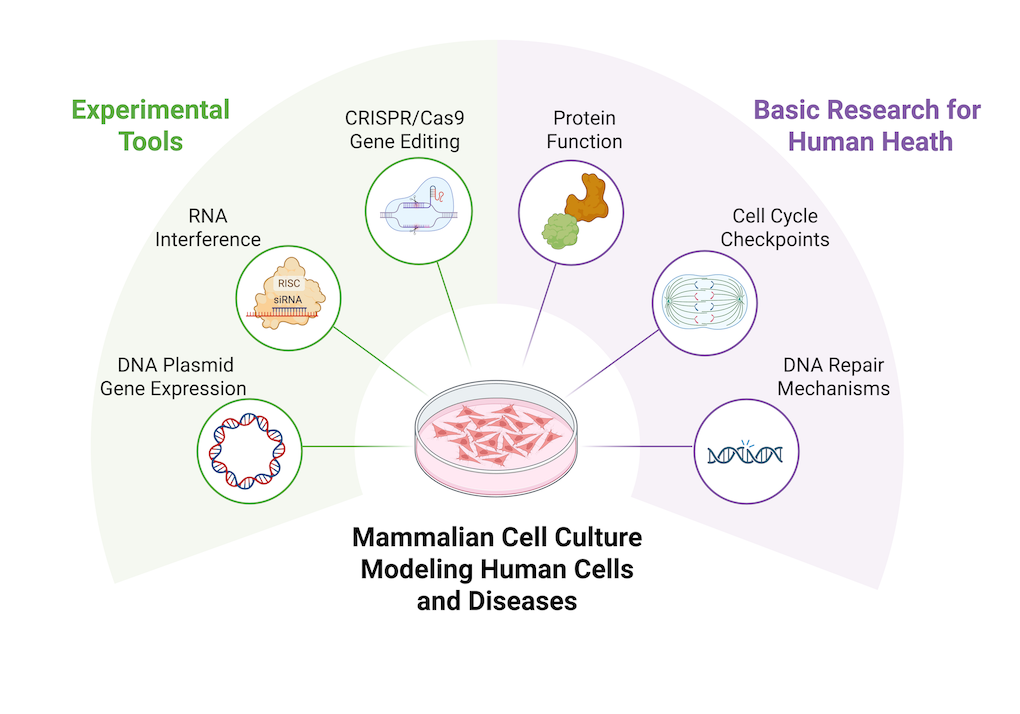
 Mammalian Cell Culture
|
 Cell Biology
|
 Cancer Biology
|
 Biochemistry
|
 Genetics
|
 DNA Repair
|
Research Educator
Genetics Homologous DNA Repair Chromosome Segregation and Aneuploidy Cell Cycle Checkpoint Mechanisms
Andrew Ziesel
Biomedical Chemistry, Research Assistant Professor
Research Interests
Dr. Andrew Ziesel is the FRI Research Educator for the Biomedical Chemistry research stream. His research focuses on understanding the biochemical mechanisms that regulate DNA damage repair and faithful chromosome segregation during cellular divisions. When these mechanisms fail, it can lead to genetic mutations and diseases, including cancer. Insights into how mammalian cells prevent disease-causing mutations can reveal new cellular pathways to target with clinical treatments. Dr. Ziesel is enthusiastic for making undergraduate research more accessible to students by removing inhibitory barriers and teaching students the universal skills needed to succeed as a scientist. Dr. Ziesel enjoys mentoring and investing in the success of his students while creating a positive and collaborative research environment.
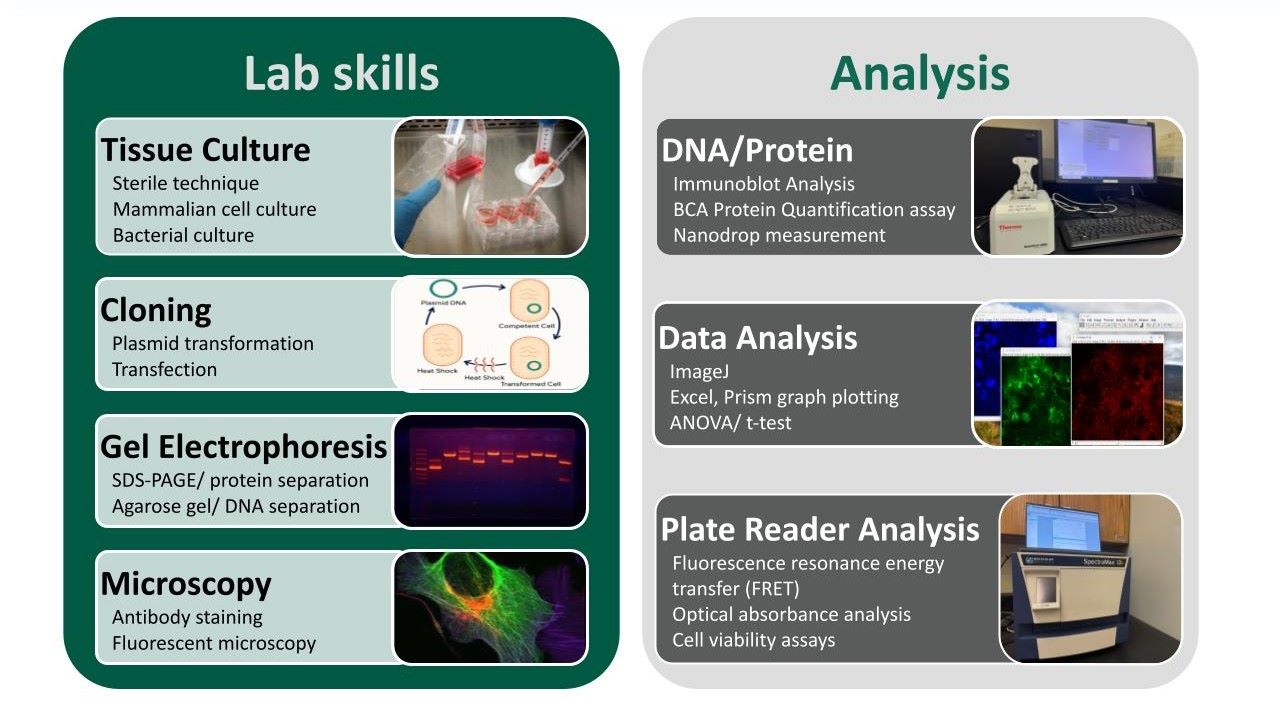
Research Techniques
Research Projects
-
Cohort 11 (2024-2025)
- Impact of polyvinyl chloride (PVC) microparticles on the intestinal epithelium
- The effects of SiO2 nanoparticles on tight junction protein Occludin formation
- The impact of polystyrene microplastics on the SLC5A6 transporter and intestinal epithelial function
- The impact of TiO2 and Ag nanoparticles on intestinal integrity
- The impact of ZnO nanoparticles on intestinal epithelial function

-
Cohort 10 (2023-2024)
- Characterization of tBT-HBT as an inhibitor and PAPP as an activator of wild-type and mutant hedgehog protein
- Determining autoprocessing levels in mutant hedgehog gene through fluorescence resonance energy transfer
- Determining the effects of the PAPP molecule on desert hedgehog (DHH) pathway
- Investigating zinc inhibition of sonic hedgehog autoprocessing and PAPP activation
- The effectiveness of tBT-HBT in inhibiting the sonic hedgehog signaling pathway

-
Cohort 9 (2022-2023)
- Detecting hSERT expression in mammalian cells upon treatment with ceftriaxone and ticarcillin
- Determining oxidative stress and cell viability of co-treatment of doxorubicin & docetaxel in lung cancer
- Investigating oxidative stress in the Bcl-2 pathway of A549 cells in the presence of docetaxel and BAY 60-6583
- Investigating the expression of hSERT in HEK cells in the presence of ceftriaxone with SSRI co-treatment
- Measuring BAX expression and cytotoxic effects of sulforaphane in cotreatment with paclitaxel in MDA cells
- Monitoring GLT-1 expression and cellular response to antibiotic treatment in kidney cells
-
Cohort 8 (2021-2022)
- Cellular oxidative stress increases upon co-treatment with panobinostat and temozolomide in the SKOV3 cell line
- Examining oxidative stress in the Bcl-2 pathway of A549 cells in the presence of a chemotherapy compound
- Expression of Anap labeled ASCT2 and beta-tubulin in HEK293T cells
- Incorporation of ncAA in human seritonin transporter (hSERT) and beta-tubulin in mammalian cells
- Monitoring oxidative stress in the presence of doxorubicin, and copper-doxorubicin in MDA-MB-231 cells
- The effects of doxorubicin and cyclophosphamide on oxidative stress in MDA-MB-231 cells
-
Cohort 7 (2020-2021)
- Determination of the third sodium binding site of GLT-1 through mutagenesis and expression in HEK cells
- Effect of doxorubicin and imatinib on oxidative stress in MDA-MB-231 breast cancer cells
- Incorporation of the ncAA, ANAP, in hSERT, expressed in a mammalian system
- Monitoring oxidative stress in the presence of taxol and etoposide chemotherapy drugs in A549 cells
- Monitoring the effects of beta-lactam antibiotics on glutamate transporter expression in HEK293T17 cells
- The effect of fluoxetine treatment on hSERT expression in a mammalian system
-
Cohort 6 (2019-2020)
- Investigating oxidative stress in A549 cells in the presence of chemotherapy agents
- Monitoring hSERT expression response to the β-Lactam antibiotic ampicillin
- Monitoring oxidative stress levels upon glycerin and propylene glycol treatment in A549 cells
- Monitoring the effect of Bak and Bax on oxidative stress in A549 cells in the presence of Trichostatin A
- Optimization toward the direct labeling of β-tubulin with ncAA within a mammalian system
-
Cohort 5 (2018-2019)
- Determining the Third Sodium Ion Coordination Site of GLT-1
- Incorporation of non-canonical amino acids into beta-tubulin
- Monitoring Oxidative Stress in A549 cells treated with Etoposide
- Monitoring Oxidative Stress in A549 cells treated with Trichostatin A
- Monitoring the Effect of Antibiotic treatment on the expression of SerotoninH6 Transporters in HEK293T17 cells
- Incorporation of unnatural amino acids into a fluorescent protein

-
Cohort 4 (2017-2018)
- EAAT2 and EAAT3 Response to the β-Lactam Antibiotics: Ceftriaxone and Amoxicillin
- Investigating Oxidative Stress in the Bcl-2 Pathway of A549 Cells in the Presence of Taxol and Colchicine
- Monitoring Lung Carcinoma Cellular Response to Artificial Sweeteners
- Monitoring Oxidative Stress in the Presence of a HDACi Chemotherapy Drug in A549 Cells
- Optimization of Monitoring Glutamate Transport and Investigating the Third Sodium Coordination Site in the GLT-Transporter

-
Cohort 3 (2016-2017)
- The Effect of DNA targeting Chemotherapy Drugs on Carbonylation in A549 cells
- Determining the Entry Mechanism of Methionine Sulfoximine into C6 Cells
- Investigating the Third Sodium binding site of the GLT-1 glutamate transporter
- Detecting Carbonylation in the Presence of Histone-targeting Chemotherapy Drugs in A549 Cells
- The Effect of Microtubule Destabilizing Chemotherapy Drugs on Carbonylation in A549 cells

-
Cohort 2 (2015-2016)
- Effects of Zn2+ and Ni2+ on glutamate transport
- Protective effects of fluoxetine and venlafaxine against oxidative stress
- Effect of beta-lactam antibiotics on EAAC1 expression and glutamate flux
- Investigating glutamate uptake activity in EAAC1
- Effect of caffeine and theophylline on cellular oxidative stress
- Investigating the antioxidant effect of acetylsalicylic acid in A549 cells with colchicine-induced oxidative stress

Research Stream Collaborators

Research Interests


Christof T. Grewer
Professor; Department Chair
Research Interests

Research Interests